Abstract
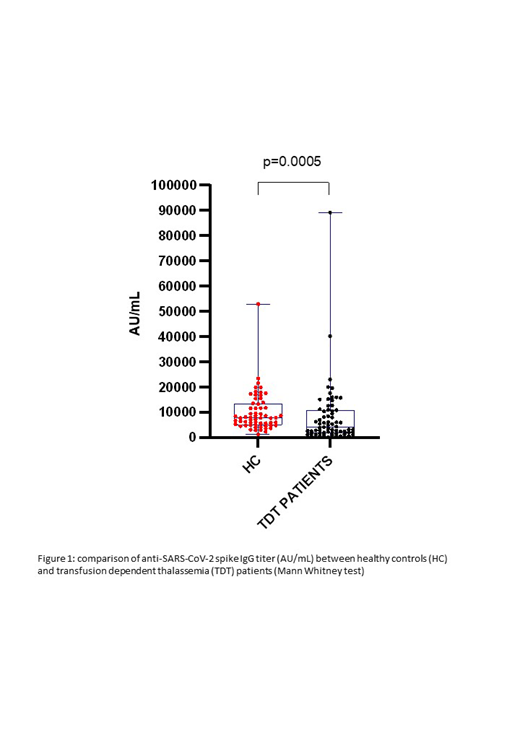
Thalassemia is an inherited blood disorder characterized by defective hemoglobin production, ineffective erythropoiesis and chronic hemolytic anemia. Patients with both transfusion-dependent thalassemia (TDT) and non-transfusion dependent thalassemia (NTDT) have risk factors associated with severe SARS-CoV-2 infection including iron overload, endocrinopathies, massive splenomegaly or previous splenectomy and coagulopathy (Motta et al, Am J Hematol, 95: E198-E199., 2020). Although vaccination is encouraged for these patients, data on the efficacy and safety of anti Sars-CoV-2 vaccines are limited (Karimi, M, et al, Br J Haematol, 190: e137-e140, 2020; Mandana Zafari, et al, Hemoglobin, 45:1, 1-4, 2021) due to exclusion of these patients from clinical trials. In a single center, prospective, cohort study we compared 67 patients affected by TDT to 61 healthy controls (HC), matched for age and sex. Study population and HC received two doses of BNT162b2 anti-SARS-Cov-2 mRNA vaccine on days 1 and 21, between April 1st and May 15 th, 2021. Serological tests were performed by a commercially available immunoassay for the quantitative determination of anti-spike IgG antibodies to SARS-CoV-2. The results were reported as Arbitrary Units (AU)/mL, with a cut-off for defining response as 50 or greater AU/ml. Patients and HC samples were collected four weeks after the second dose of vaccine. Median age of patients was 43 years (range 19-77), 39% of them were male and 61% were female. Median age of HC was 39 years (range 19-86), 43% of them were male and 57% were female. All controls achieved a response (50 or greater AU/mL) to vaccination, whereas 66/67 (98,5%) patients responded. Antibody titers were significantly higher (p=0.0005) in the HC group (mean 9863 ± 7784; median 7712, range 1206-51664) compared to patients (mean 7945 ± 12326; median 4025, range 19-89202) (Figure 1). When analyzing the patients' factors, age, sex, transfusion interval, serum ferritin level, and spleen size did not impact on the response to vaccination. With a median follow-up of 12 weeks, no relevant side effects were recorded after vaccination and no case of COVID19 occurred among vaccinated TDT patients. In conclusion, BNT162b2 anti-SARS-Cov-2 mRNA vaccine demonstrated efficacy and safety in our cohort of TDT patients. Response rate was similar to that of HC. Nevertheless, antibody titers in TDT patients were significantly lower than in HC. Further observations are ongoing to assess duration of response, efficacy and possible factors influencing this finding.
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal